What 3 Gases Makeup Our Atsophere

NASA photo showing Earth'south atmosphere at sunset, with Globe silhouetted

Blue light is scattered more than than other wavelengths by the gases in the atmosphere, surrounding Globe in a visibly blue layer when seen from space on lath the ISS at an altitude of 335 km (208 mi).[i]

Composition of Earth's atmosphere by molecular count, excluding water vapor. Lower pie represents trace gases that together compose about 0.0434% of the atmosphere (0.0442% at August 2021 concentrations[2] [iii]). Numbers are mainly from 2000, with CO2 and methane from 2019, and exercise not represent any unmarried source.[4]
The atmosphere of Globe, commonly known as air, is the layer of gases retained by Earth's gravity that surrounds the planet and forms its planetary atmosphere. The temper of Earth protects life on World by creating force per unit area allowing for liquid h2o to exist on the Earth'due south surface, absorbing ultraviolet solar radiation, warming the surface through heat memory (greenhouse effect), and reducing temperature extremes between day and night (the diurnal temperature variation).
By mole fraction (i.e., past number of molecules), dry air contains 78.08% nitrogen, xx.95% oxygen, 0.93% argon, 0.04% carbon dioxide, and small amounts of other gases.[viii] Air as well contains a variable amount of water vapor, on average effectually 1% at sea level, and 0.4% over the entire atmosphere. Air composition, temperature, and atmospheric pressure vary with altitude. Within the atmosphere, air suitable for utilise in photosynthesis past terrestrial plants and animate of terrestrial animals is found only in Earth's troposphere.[ citation needed ]
Earth'southward early on temper consisted of gases in the solar nebula, primarily hydrogen. The atmosphere changed significantly over time, affected by many factors such every bit volcanism, life, and weathering. Recently, human activity has also contributed to atmospheric changes, such equally global warming, ozone depletion and acid deposition.
The temper has a mass of virtually 5.15×10 18 kg,[9] iii quarters of which is within near 11 km (6.8 mi; 36,000 ft) of the surface. The atmosphere becomes thinner with increasing altitude, with no definite boundary between the atmosphere and outer space. The Kármán line, at 100 km (62 mi) or one.57% of World's radius, is oft used as the border between the atmosphere and outer space. Atmospheric effects become noticeable during atmospheric reentry of spacecraft at an altitude of around 120 km (75 mi). Several layers tin be distinguished in the atmosphere, based on characteristics such equally temperature and composition.
The study of Earth'south atmosphere and its processes is called atmospheric science (aerology), and includes multiple subfields, such as climatology and atmospheric physics. Early pioneers in the field include Léon Teisserenc de Bort and Richard Assmann.[10] The study of celebrated atmosphere is called paleoclimatology.
Limerick

Hateful atmospheric water vapor
The 3 major constituents of Earth'due south atmosphere are nitrogen, oxygen, and argon. Water vapor accounts for roughly 0.25% of the atmosphere by mass. The concentration of water vapor (a greenhouse gas) varies significantly from effectually 10 ppm by mole fraction in the coldest portions of the atmosphere to as much every bit 5% past mole fraction in hot, humid air masses, and concentrations of other atmospheric gases are typically quoted in terms of dry out air (without water vapor).[11] : 8 The remaining gases are oftentimes referred to every bit trace gases,[12] among which are other greenhouse gases, principally carbon dioxide, marsh gas, nitrous oxide, and ozone. As well argon, already mentioned, other noble gases, neon, helium, krypton, and xenon are also present. Filtered air includes trace amounts of many other chemical compounds. Many substances of natural origin may be nowadays in locally and seasonally variable pocket-size amounts every bit aerosols in an unfiltered air sample, including dust of mineral and organic limerick, pollen and spores, sea spray, and volcanic ash. Various industrial pollutants as well may be present as gases or aerosols, such as chlorine (elemental or in compounds), fluorine compounds and elemental mercury vapor. Sulfur compounds such every bit hydrogen sulfide and sulfur dioxide (Sotwo) may be derived from natural sources or from industrial air pollution.
| Gas | Mole fraction (A) | ||
|---|---|---|---|
| Name | Formula | in ppm (B) | in % |
| Nitrogen | Nii | 780,840 | 78.084 |
| Oxygen | O2 | 209,460 | 20.946 |
| Argon | Ar | 9,340 | 0.9340 |
| Carbon dioxide (April, 2022)(C) [13] | CO2 | 417 | 0.0417 |
| Neon | Ne | 18.xviii | 0.001818 |
| Helium | He | 5.24 | 0.000524 |
| Methane | CH4 | 1.87 | 0.000187 |
| Krypton | Kr | 1.14 | 0.000114 |
| Not included in above dry atmosphere: | |||
| Water vapor(D) | HiiO | 0–30,000(D) | 0–3%(E) |
notes:
| |||
The boilerplate molecular weight of dry air, which tin can be used to calculate densities or to convert betwixt mole fraction and mass fraction, is nearly 28.946[14] or 28.96[15] [16] g/mol. This is decreased when the air is humid.
The relative concentration of gases remains constant until almost 10,000 k (33,000 ft).[17]

The mole fraction of the primary constituents of the Earth'southward atmosphere every bit a office of height according to the MSIS-Eastward-90 atmospheric model.
Stratification

Earth's atmosphere Lower four layers of the atmosphere in iii dimensions as seen diagonally from higher up the exobase. Layers drawn to scale, objects within the layers are not to scale. Aurorae shown here at the lesser of the thermosphere tin actually course at whatever altitude in this atmospheric layer.
[18]In general, air pressure and density subtract with altitude in the temper. Still, the temperature has a more complicated profile with distance, and may remain relatively constant or even increase with altitude in some regions (run across the temperature section, below). Because the general pattern of the temperature/distance contour, or lapse rate, is constant and measurable past means of instrumented balloon soundings, the temperature behavior provides a useful metric to distinguish atmospheric layers. In this way, Earth'southward atmosphere tin can be divided (called atmospheric stratification) into five primary layers: troposphere, stratosphere, mesosphere, thermosphere, and exosphere.[19] The altitudes of the five layers are as follows:
- Exosphere: 700 to 10,000 km (440 to 6,200 miles)[20]
- Thermosphere: eighty to 700 km (50 to 440 miles)[21]
- Mesosphere: 50 to 80 km (31 to 50 miles)
- Stratosphere: 12 to 50 km (7 to 31 miles)
- Troposphere: 0 to 12 km (0 to vii miles)[22]
Exosphere
The exosphere is the outermost layer of Globe's atmosphere (i.e. the upper limit of the atmosphere). It extends from the thermopause, at the pinnacle of the thermosphere at an altitude of about 700 km in a higher place sea level, to about ten,000 km (6,200 mi; 33,000,000 ft), where it merges into the solar air current.[20]
This layer is mainly composed of extremely low densities of hydrogen, helium and several heavier molecules including nitrogen, oxygen and carbon dioxide closer to the exobase. The atoms and molecules are so far apart that they can travel hundreds of kilometers without colliding with one another. Thus, the exosphere no longer behaves like a gas, and the particles constantly escape into infinite. These free-moving particles follow ballistic trajectories and may migrate in and out of the magnetosphere or the solar air current.
The exosphere is as well far in a higher place Earth for meteorological phenomena to be possible. All the same, Globe's auroras—the aurora borealis (northern lights) and aurora australis (southern lights)—sometimes occur in the lower office of the exosphere, where they overlap into the thermosphere. The exosphere contains many of the artificial satellites that orbit Earth.
Thermosphere
The thermosphere is the second-highest layer of Earth's atmosphere. It extends from the mesopause (which separates information technology from the mesosphere) at an distance of about 80 km (l mi; 260,000 ft) up to the thermopause at an distance range of 500–thousand km (310–620 mi; 1,600,000–3,300,000 ft). The peak of the thermopause varies considerably due to changes in solar activity.[21] Considering the thermopause lies at the lower boundary of the exosphere, it is also referred to every bit the exobase. The lower part of the thermosphere, from 80 to 550 kilometres (50 to 342 mi) in a higher place Globe's surface, contains the ionosphere.
The temperature of the thermosphere gradually increases with height and can ascent as high as 1500 °C (2700 °F), though the gas molecules are and so far apart that its temperature in the usual sense is not very meaningful. The air is so rarefied that an individual molecule (of oxygen, for case) travels an average of 1 kilometre (0.62 mi; 3300 ft) between collisions with other molecules.[23] Although the thermosphere has a high proportion of molecules with high free energy, information technology would not feel hot to a human in direct contact, considering its density is as well depression to conduct a pregnant amount of energy to or from the skin.
This layer is completely cloudless and free of water vapor. However, non-hydrometeorological phenomena such as the aurora borealis and aurora australis are occasionally seen in the thermosphere. The International Space Station orbits in this layer, betwixt 350 and 420 km (220 and 260 mi). It is this layer where many of the satellites orbiting the earth are present.
Mesosphere
The mesosphere is the third highest layer of Earth's atmosphere, occupying the region above the stratosphere and beneath the thermosphere. It extends from the stratopause at an distance of about 50 km (31 mi; 160,000 ft) to the mesopause at 80–85 km (50–53 mi; 260,000–280,000 ft) above bounding main level.
Temperatures drop with increasing altitude to the mesopause that marks the top of this centre layer of the atmosphere. It is the coldest place on Earth and has an boilerplate temperature around −85 °C (−120 °F; 190 K).[24] [25]
Just below the mesopause, the air is so cold that fifty-fifty the very scarce h2o vapor at this altitude can sublimate into polar-mesospheric noctilucent clouds of ice particles. These are the highest clouds in the atmosphere and may be visible to the naked eye if sunlight reflects off them about an hour or ii after dusk or similarly before sunrise. They are most readily visible when the Sun is effectually 4 to 16 degrees beneath the horizon. Lightning-induced discharges known as transient luminous events (TLEs) occasionally form in the mesosphere above tropospheric thunderclouds. The mesosphere is also the layer where about meteors burn up upon atmospheric entrance. It is too loftier higher up Earth to be accessible to jet-powered aircraft and balloons, and too low to permit orbital spacecraft. The mesosphere is mainly accessed by sounding rockets and rocket-powered shipping.
Stratosphere
The stratosphere is the second-lowest layer of World'southward atmosphere. Information technology lies higher up the troposphere and is separated from it by the tropopause. This layer extends from the top of the troposphere at roughly 12 km (7.five mi; 39,000 ft) above World's surface to the stratopause at an altitude of about 50 to 55 km (31 to 34 mi; 164,000 to 180,000 ft).
The atmospheric pressure level at the height of the stratosphere is roughly 1/1000 the pressure level at sea level. It contains the ozone layer, which is the part of Earth's atmosphere that contains relatively high concentrations of that gas. The stratosphere defines a layer in which temperatures rise with increasing distance. This ascent in temperature is caused by the absorption of ultraviolet radiation (UV) radiation from the Sunday by the ozone layer, which restricts turbulence and mixing. Although the temperature may be −sixty °C (−76 °F; 210 K) at the tropopause, the elevation of the stratosphere is much warmer, and may be about 0 °C.[26]
The stratospheric temperature profile creates very stable atmospheric atmospheric condition, so the stratosphere lacks the conditions-producing air turbulence that is so prevalent in the troposphere. Consequently, the stratosphere is well-nigh completely free of clouds and other forms of weather. However, polar stratospheric or nacreous clouds are occasionally seen in the lower part of this layer of the atmosphere where the air is coldest. The stratosphere is the highest layer that can be accessed by jet-powered aircraft.
Troposphere
The troposphere is the everyman layer of Earth's temper. It extends from Earth'due south surface to an average height of about 12 km (vii.five mi; 39,000 ft), although this altitude varies from most 9 km (5.6 mi; 30,000 ft) at the geographic poles to 17 km (11 mi; 56,000 ft) at the Equator,[22] with some variation due to weather. The troposphere is divisional above by the tropopause, a purlieus marked in most places by a temperature inversion (i.e. a layer of relatively warm air above a colder ane), and in others by a zone that is isothermal with top.[27] [28]
Although variations do occur, the temperature usually declines with increasing altitude in the troposphere because the troposphere is more often than not heated through free energy transfer from the surface. Thus, the lowest function of the troposphere (i.e. Earth's surface) is typically the warmest section of the troposphere. This promotes vertical mixing (hence, the origin of its name in the Greek word τρόπος, tropos, meaning "turn"). The troposphere contains roughly 80% of the mass of Earth'southward atmosphere.[29] The troposphere is denser than all its overlying layers because a larger atmospheric weight sits on acme of the troposphere and causes information technology to be well-nigh severely compressed. 50 pct of the total mass of the temper is located in the lower 5.6 km (3.5 mi; 18,000 ft) of the troposphere.
Near all atmospheric h2o vapor or wet is found in the troposphere, then it is the layer where most of World'southward weather takes place. It has basically all the atmospheric condition-associated cloud genus types generated by active wind circulation, although very tall cumulonimbus thunder clouds can penetrate the tropopause from below and rise into the lower part of the stratosphere. Most conventional aviation activeness takes place in the troposphere, and it is the only layer that tin can be accessed by propeller-driven aircraft.

Space Shuttle Endeavour orbiting in the thermosphere. Because of the angle of the photo, it appears to straddle the stratosphere and mesosphere that actually lie more than 250 km (160 mi) below. The orange layer is the troposphere, which gives manner to the whitish stratosphere and then the blue mesosphere.[30]
Other layers
Within the five chief layers to a higher place, which are largely determined past temperature, several secondary layers may be distinguished by other properties:
- The ozone layer is contained within the stratosphere. In this layer ozone concentrations are nearly 2 to 8 parts per one thousand thousand, which is much higher than in the lower atmosphere simply notwithstanding very small compared to the main components of the atmosphere. It is mainly located in the lower portion of the stratosphere from near xv–35 km (9.iii–21.7 mi; 49,000–115,000 ft), though the thickness varies seasonally and geographically. About 90% of the ozone in Earth's atmosphere is contained in the stratosphere.
- The ionosphere is a region of the atmosphere that is ionized by solar radiation. Information technology is responsible for auroras. During daytime hours, it stretches from 50 to 1,000 km (31 to 621 mi; 160,000 to 3,280,000 ft) and includes the mesosphere, thermosphere, and parts of the exosphere. Nevertheless, ionization in the mesosphere largely ceases during the dark, so auroras are usually seen only in the thermosphere and lower exosphere. The ionosphere forms the inner border of the magnetosphere. It has applied importance considering it influences, for example, radio propagation on World.
- The homosphere and heterosphere are defined by whether the atmospheric gases are well mixed. The surface-based homosphere includes the troposphere, stratosphere, mesosphere, and the lowest part of the thermosphere, where the chemic composition of the atmosphere does non depend on molecular weight because the gases are mixed by turbulence.[31] This relatively homogeneous layer ends at the turbopause establish at about 100 km (62 mi; 330,000 ft), the very border of infinite itself as accepted past the FAI, which places it about 20 km (12 mi; 66,000 ft) above the mesopause.
- Above this altitude lies the heterosphere, which includes the exosphere and most of the thermosphere. Here, the chemical composition varies with altitude. This is because the distance that particles can move without colliding with one another is large compared with the size of motions that cause mixing. This allows the gases to stratify by molecular weight, with the heavier ones, such every bit oxygen and nitrogen, present only about the bottom of the heterosphere. The upper part of the heterosphere is composed almost completely of hydrogen, the lightest chemical element.[ description needed ]
- The planetary boundary layer is the role of the troposphere that is closest to World'south surface and is directly affected by information technology, mainly through turbulent diffusion. During the 24-hour interval the planetary boundary layer usually is well-mixed, whereas at night it becomes stably stratified with weak or intermittent mixing. The depth of the planetary boundary layer ranges from as piffling as about 100 metres (330 ft) on clear, calm nights to iii,000 m (9,800 ft) or more during the afternoon in dry regions.
The average temperature of the atmosphere at Earth'south surface is xiv °C (57 °F; 287 One thousand)[32] or fifteen °C (59 °F; 288 K),[33] depending on the reference.[34] [35] [36]
Physical properties
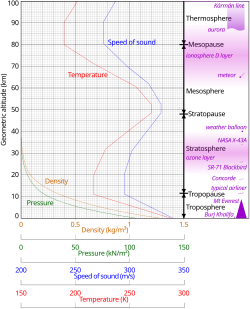
Force per unit area and thickness
The average atmospheric pressure level at sea level is defined by the International Standard Atmosphere as 101325 pascals (760.00 Torr; fourteen.6959 psi; 760.00 mmHg). This is sometimes referred to every bit a unit of measurement of standard atmospheres (atm). Total atmospheric mass is five.1480×1018 kg (1.135×xxix lb),[38] about 2.5% less than would be inferred from the average sea level pressure and Globe's expanse of 51007.2 megahectares, this portion being displaced past Globe's mountainous terrain. Atmospheric pressure is the full weight of the air above unit area at the point where the pressure is measured. Thus air pressure varies with location and conditions.
If the entire mass of the temper had a uniform density equal to bounding main level density (about ane.2 kg per m3) from sea level upwards, it would finish abruptly at an altitude of 8.50 km (27,900 ft).
Air pressure really decreases exponentially with altitude, dropping past half every 5.6 km (18,000 ft) or by a gene of ane/east (0.368) every 7.64 km (25,100 ft), (this is called the scale height) -- for altitudes out to around seventy km (43 mi; 230,000 ft). However, the atmosphere is more accurately modeled with a customized equation for each layer that takes gradients of temperature, molecular composition, solar radiation and gravity into account. At heights over 100 km, an atmosphere may no longer exist well mixed. So each chemical species has its own scale meridian.
In summary, the mass of Globe's atmosphere is distributed approximately every bit follows:[39]
- 50% is below v.half dozen km (eighteen,000 ft).
- 90% is below xvi km (52,000 ft).
- 99.99997% is below 100 km (62 mi; 330,000 ft), the Kármán line. By international convention, this marks the beginning of space where human travelers are considered astronauts.
By comparing, the summit of Mt. Everest is at 8,848 m (29,029 ft); commercial airliners typically prowl betwixt ten and 13 km (33,000 and 43,000 ft) where the lower density and temperature of the air improve fuel economy; weather balloons accomplish xxx.four km (100,000 ft) and above; and the highest Ten-xv flight in 1963 reached 108.0 km (354,300 ft).
Fifty-fifty higher up the Kármán line, significant atmospheric furnishings such every bit auroras still occur. Meteors begin to glow in this region, though the larger ones may not burn upward until they penetrate more than securely. The various layers of Globe's ionosphere, important to HF radio propagation, begin beneath 100 km and extend beyond 500 km. By comparison, the International Space Station and Space Shuttle typically orbit at 350–400 km, within the F-layer of the ionosphere where they encounter plenty atmospheric drag to require reboosts every few months, otherwise, orbital decay will occur resulting in a return to Earth. Depending on solar activity, satellites tin experience noticeable atmospheric drag at altitudes every bit high equally 700–800 km.
Temperature
The division of the temper into layers mostly past reference to temperature is discussed above. Temperature decreases with distance starting at sea level, merely variations in this trend begin above xi km, where the temperature stabilizes over a large vertical distance through the rest of the troposphere. In the stratosphere, starting in a higher place about 20 km, the temperature increases with pinnacle, due to heating within the ozone layer caused by the capture of pregnant ultraviolet radiation from the Sun past the dioxygen and ozone gas in this region. Still another region of increasing temperature with altitude occurs at very high altitudes, in the aptly-named thermosphere above 90 km.
Speed of sound
Because in an platonic gas of abiding composition the speed of sound depends only on temperature and not on pressure or density, the speed of sound in the temper with altitude takes on the class of the complicated temperature profile (see illustration to the right), and does not mirror altitudinal changes in density or force per unit area.
Density and mass

Temperature and mass density against distance from the NRLMSISE-00 standard temper model (the eight dotted lines in each "decade" are at the viii cubes 8, 27, 64, ..., 729)
The density of air at body of water level is about one.2 kg/thousand3 (1.2 g/50, 0.0012 g/cm3). Density is non measured directly merely is calculated from measurements of temperature, pressure and humidity using the equation of country for air (a form of the ideal gas law). Atmospheric density decreases as the altitude increases. This variation can be approximately modeled using the barometric formula. More sophisticated models are used to predict the orbital decay of satellites.
The average mass of the atmosphere is about five quadrillion (5×10 15 ) tonnes or 1/1,200,000 the mass of Earth. According to the American National Center for Atmospheric Inquiry, "The full mean mass of the atmosphere is 5.1480×10 18 kg with an annual range due to h2o vapor of i.2 or i.5×ten 15 kg, depending on whether surface pressure level or h2o vapor data are used; somewhat smaller than the previous estimate. The mean mass of h2o vapor is estimated equally i.27×10 16 kg and the dry air mass as 5.1352 ±0.0003×10 18 kg."
Optical backdrop
Solar radiation (or sunlight) is the energy Earth receives from the Sunday. Earth too emits radiation back into space, but at longer wavelengths that humans cannot see. Function of the incoming and emitted radiation is captivated or reflected by the atmosphere. In May 2017, glints of light, seen as twinkling from an orbiting satellite a 1000000 miles away, were constitute to be reflected low-cal from ice crystals in the atmosphere.[41] [42]
Scattering
When light passes through Earth's atmosphere, photons collaborate with it through scattering. If the light does not interact with the temper, information technology is called straight radiation and is what you encounter if you lot were to await directly at the Lord's day. Indirect radiation is light that has been scattered in the atmosphere. For example, on an overcast day when you cannot see your shadow, there is no directly radiations reaching you lot, it has all been scattered. Equally another case, due to a phenomenon called Rayleigh scattering, shorter (blue) wavelengths scatter more easily than longer (scarlet) wavelengths. This is why the sky looks blue; you are seeing scattered blue low-cal. This is also why sunsets are cherry. Because the Lord's day is close to the horizon, the Sun'due south rays pass through more than atmosphere than normal before reaching your heart. Much of the blue light has been scattered out, leaving the red light in a sunset.
Absorption
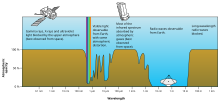
Rough plot of World'due south atmospheric transmittance (or opacity) to various wavelengths of electromagnetic radiations, including visible light.
Different molecules absorb different wavelengths of radiations. For example, O2 and Oiii blot almost all radiations with wavelengths shorter than 300 nanometers. Water (H2O) absorbs at many wavelengths higher up 700 nm. When a molecule absorbs a photon, information technology increases the energy of the molecule. This heats the atmosphere, but the temper also cools past emitting radiation, as discussed below.
The combined assimilation spectra of the gases in the atmosphere leave "windows" of low opacity, assuasive the manual of just certain bands of calorie-free. The optical window runs from around 300 nm (ultraviolet-C) up into the range humans can see, the visible spectrum (usually called low-cal), at roughly 400–700 nm and continues to the infrared to effectually 1100 nm. There are also infrared and radio windows that transmit some infrared and radio waves at longer wavelengths. For case, the radio window runs from about ane centimeter to well-nigh eleven-meter waves.
Emission
Emission is the reverse of assimilation, it is when an object emits radiation. Objects tend to emit amounts and wavelengths of radiations depending on their "black trunk" emission curves, therefore hotter objects tend to emit more than radiations, with shorter wavelengths. Colder objects emit less radiation, with longer wavelengths. For example, the Sunday is approximately six,000 K (5,730 °C; x,340 °F), its radiation peaks near 500 nm, and is visible to the human eye. Globe is approximately 290 G (17 °C; 62 °F), and then its radiations peaks nearly 10,000 nm, and is much likewise long to be visible to humans.
Considering of its temperature, the atmosphere emits infrared radiation. For example, on clear nights Globe'south surface cools down faster than on cloudy nights. This is considering clouds (H2O) are strong absorbers and emitters of infrared radiation. This is besides why information technology becomes colder at dark at higher elevations.
The greenhouse effect is direct related to this absorption and emission consequence. Some gases in the temper absorb and emit infrared radiation, simply do non collaborate with sunlight in the visible spectrum. Common examples of these are CO2 and H2O.
Refractive alphabetize

The refractive index of air is close to, but only greater than 1. Systematic variations in the refractive alphabetize can lead to the bending of light rays over long optical paths. 1 example is that, under some circumstances, observers onboard ships tin can run into other vessels just over the horizon because low-cal is refracted in the same direction as the curvature of Globe's surface.
The refractive index of air depends on temperature,[43] giving rise to refraction effects when the temperature gradient is big. An instance of such effects is the mirage.
Circulation
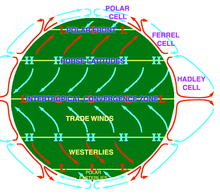
An idealised view of three pairs of big apportionment cells.
Atmospheric circulation is the large-scale movement of air through the troposphere, and the means (with bounding main apportionment) past which oestrus is distributed around World. The large-scale structure of the atmospheric circulation varies from yr to twelvemonth, only the basic structure remains adequately constant considering information technology is adamant by Earth'south rotation rate and the divergence in solar radiation between the equator and poles.
Development of Earth'south temper
Primeval atmosphere
The offset temper consisted of gases in the solar nebula, primarily hydrogen. There were probably simple hydrides such equally those now found in the gas giants (Jupiter and Saturn), notably water vapor, marsh gas and ammonia.[44]
Second atmosphere
Outgassing from volcanism, supplemented by gases produced during the belatedly heavy bombardment of World by huge asteroids, produced the next atmosphere, consisting largely of nitrogen plus carbon dioxide and inert gases.[44] A major role of carbon-dioxide emissions dissolved in water and reacted with metals such as calcium and magnesium during weathering of crustal rocks to class carbonates that were deposited equally sediments. Water-related sediments have been establish that engagement from as early on as 3.viii billion years ago.[45]
About three.4 billion years ago, nitrogen formed the major part of the then stable "2nd atmosphere". The influence of life has to be taken into business relationship rather soon in the history of the atmosphere because hints of early life-forms appear as early as 3.v billion years ago.[46] How Earth at that time maintained a climate warm enough for liquid water and life, if the early on Lord's day put out 30% lower solar radiance than today, is a puzzle known equally the "faint young Sunday paradox".
The geological record however shows a continuous relatively warm surface during the complete early temperature tape of Earth – with the exception of i common cold glacial stage most 2.4 billion years ago. In the late Archean Eon an oxygen-containing atmosphere began to develop, apparently produced by photosynthesizing cyanobacteria (run across Great Oxygenation Result), which have been found as stromatolite fossils from 2.7 billion years ago. The early on bones carbon isotopy (isotope ratio proportions) strongly suggests conditions similar to the current, and that the fundamental features of the carbon wheel became established as early on as 4 billion years ago.
Aboriginal sediments in the Gabon dating from between most 2.15 and two.08 billion years ago provide a record of Earth'southward dynamic oxygenation evolution. These fluctuations in oxygenation were probable driven past the Lomagundi carbon isotope circuit.[47]
Third atmosphere
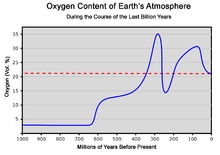
Oxygen content of the atmosphere over the last billion years[48] [49]
The abiding re-arrangement of continents by plate tectonics influences the long-term evolution of the atmosphere by transferring carbon dioxide to and from large continental carbonate stores. Costless oxygen did non exist in the atmosphere until about 2.four billion years agone during the Keen Oxygenation Event and its appearance is indicated by the end of the banded fe formations.
Before this time, any oxygen produced by photosynthesis was consumed by the oxidation of reduced materials, notably atomic number 26. Free oxygen molecules did non start to accumulate in the atmosphere until the rate of production of oxygen began to exceed the availability of reducing materials that removed oxygen. This point signifies a shift from a reducing temper to an oxidizing atmosphere. O2 showed major variations until reaching a steady state of more than fifteen% by the finish of the Precambrian.[fifty] The following time span from 539 million years ago to the present day is the Phanerozoic Eon, during the earliest menses of which, the Cambrian, oxygen-requiring metazoan life forms began to appear.
The corporeality of oxygen in the atmosphere has fluctuated over the last 600 million years, reaching a peak of about 30% effectually 280 meg years ago, significantly higher than today'south 21%. Two principal processes govern changes in the atmosphere: Plants using carbon dioxide from the atmosphere and releasing oxygen, then plants using some oxygen at night by the procedure of photorespiration while the remaining oxygen is used to suspension down organic material. Breakdown of pyrite and volcanic eruptions release sulfur into the temper, which reacts with oxygen and hence reduces its amount in the atmosphere. Nonetheless, volcanic eruptions also release carbon dioxide, which plants can convert to oxygen. The cause of the variation of the amount of oxygen in the atmosphere is non known. Periods with much oxygen in the atmosphere are associated with the rapid development of animals considering oxygen is the high-free energy molecule needed to power all complex life-forms.[51] Today's atmosphere contains 21% oxygen, which is keen enough for this rapid evolution of animals.[52]
Air pollution
Air pollution is the introduction into the temper of chemicals, particulate matter or biological materials that cause harm or discomfort to organisms.[53] Stratospheric ozone depletion is caused by air pollution, chiefly from chlorofluorocarbons and other ozone-depleting substances.
Since 1750, homo activeness has increased the concentrations various greenhouse gases, virtually importantly carbon dioxide, methane and nitrous oxide. This increase has caused an observed rise in global temperatures. Global boilerplate surface temperatures were 1.1 °C college in the 2011-2020 decade than they were in 1850.[54]
Animation shows the buildup of tropospheric CO2 in the Northern Hemisphere with a maximum around May. The maximum in the vegetation cycle follows in the late summer. Following the height in vegetation, the drawdown of atmospheric CO2 due to photosynthesis is apparent, particularly over the boreal forests.
Images from infinite
On Oct 19, 2015, NASA started a website containing daily images of the full sunlit side of Earth at https://epic.gsfc.nasa.gov/. The images are taken from the Deep Infinite Climate Observatory (DSCOVR) and show Earth as information technology rotates during a day.[55]
-
Limb view, of Earth's atmosphere. Colors roughly denote the layers of the atmosphere.
-
This image shows the Moon at the centre, with the limb of Earth almost the lesser transitioning into the orange-colored troposphere. The troposphere ends abruptly at the tropopause, which appears in the prototype every bit the sharp purlieus between the orange- and blue-colored temper. The silver-blue noctilucent clouds extend far higher up Earth'south troposphere.
Meet also
- Aerial perspective
- Air (classical element)
- Air glow
- Airshed
- Atmospheric dispersion modeling
- Atmospheric electricity
- Atmospheric Radiations Measurement Climate Inquiry Facility (ARM) (in the U.S.)
- Atmospheric stratification
- Biosphere
- Climate system
- Earth'south energy budget
- COSPAR international reference atmosphere (CIRA)
- Environmental bear upon of aviation
- Global dimming
- Historical temperature tape
- Hydrosphere
- Hypermobility (travel)
- Kyoto Protocol
- Leaching (agronomics)
- Lithosphere
- Reference atmospheric model
References
- ^ "Gateway to Astonaut Photos of Earth". NASA. Retrieved 2018-01-29 .
- ^ a b "Trends in Atmospheric Carbon Dioxide", Global Greenhouse Gas Reference Network, NOAA, 2019, retrieved 2019-05-31
- ^ a b "Trends in Atmospheric Methyl hydride", Global Greenhouse Gas Reference Network, NOAA, 2019, retrieved 2019-05-31
- ^ a b Haynes, H. M., ed. (2016–2017), CRC Handbook of Chemical science and Physics (97th ed.), CRC Press, p. 14-iii, ISBN978-1-4987-5428-six , which cites Allen's Astrophysical Quantities but includes only 10 of its largest constituents.
- ^ Cox, Arthur N., ed. (2000), Allen'south Astrophysical Quantities (Fourth ed.), AIP Press, pp. 258–259, ISBN0-387-98746-0 , which rounds N2 and Otwo to four significant digits without affecting the full because 0.004% was removed from Northward2 and added to O2. It includes xx constituents.
- ^ National Aeronautics and Space Assistants (1976), U.S. Standard Atmosphere, 1976 (PDF), p. iii
- ^ Allen, C. Westward. (1976), Astrophysical Quantities (3rd ed.), Athlone Press, p. 119, ISBN0-485-11150-0
- ^ a b Ii recent reliable sources cited here have total atmospheric compositions, including trace molecules, that exceed 100%. They are Allen's Astrophysical Quantities [5] (2000, 100.001241343%) and CRC Handbook of Chemistry and Physics [four] (2016–2017, 100.004667%), which cites Allen's Astrophysical Quantities. Both are used as references in this commodity. Both exceed 100% because their CO2 values were increased to 345 ppmv, without irresolute their other constituents to compensate. This is made worse by the April 2019 COtwo value, which is 413.32 ppmv.[ii] Although modest, the January 2019 value for CH4 is 1866.1 ppbv (parts per billion).[iii] Two older reliable sources accept dry atmospheric compositions, including trace molecules, that full less than 100%: U.Due south. Standard Atmosphere, 1976 [6] (99.9997147%); and Astrophysical Quantities [7] (1976, 99.9999357%).
- ^ Lide, David R. Handbook of Chemistry and Physics. Boca Raton, FL: CRC, 1996: 14–17
- ^ Vázquez, 1000.; Hanslmeier, A. (2006). "Historical Introduction". Ultraviolet Radiation in the Solar System. Astrophysics and Space Science Library. Vol. 331. Springer Science & Business concern Media. p. 17. Bibcode:2005ASSL..331.....V. doi:10.1007/one-4020-3730-9_1. ISBN978-ane-4020-3730-6.
- ^ a b Wallace, John Thou. and Peter V. Hobbs. Atmospheric Science: An Introductory Survey Archived 2018-07-28 at the Wayback Machine. Elsevier. 2nd Edition, 2006. ISBN 978-0-12-732951-2. Chapter 1
- ^ "Trace Gases". Ace.mmu.ac.uk. Archived from the original on 9 October 2010. Retrieved 2010-10-16 .
- ^ "Vital signs: Carbon Dioxide". NASA Climate. April 2022. Retrieved 16 May 2022.
- ^ Detlev Möller: Luft: Chemie, Physik, Biologie, Reinhaltung, Recht. Walter de Gruyter, 2003, ISBN 3-xi-016431-0, Southward. 173. (View in Google Books).
- ^ Yunus Çengel. Termodinamica due east trasmissione del calore.
- ^ "Air - Molecular Weight and Composition". www.engineeringtoolbox.com . Retrieved 2021-04-27 .
- ^ "Air Composition". The Applied science ToolBox. Retrieved 2017-07-04 .
The composition of air is unchanged until meridian of approximately x.000 m
- ^ "Exosphere - overview". UCAR. 2011. Archived from the original on 17 May 2017. Retrieved April 19, 2015.
- ^ Zell, Holly (2015-03-02). "Earth's Upper Atmosphere". NASA . Retrieved 2017-02-20 .
- ^ a b "Exosphere - overview". UCAR. 2011. Archived from the original on 17 May 2017. Retrieved April xix, 2015.
- ^ a b Randy Russell (2008). "The Thermosphere". Retrieved 2013-10-18 .
- ^ a b "The meridian of the tropopause". Das.uwyo.edu. Retrieved 2012-04-18 .
- ^ Ahrens, C. Donald. Essentials of Meteorology. Published past Thomson Brooks/Cole, 2005.
- ^ States, Robert J.; Gardner, Chester South. (January 2000). "Thermal Structure of the Mesopause Region (80–105 km) at 40°N Latitude. Role I: Seasonal Variations". Journal of the Atmospheric Sciences. 57 (1): 66–77. Bibcode:2000JAtS...57...66S. doi:10.1175/1520-0469(2000)057<0066:TSOTMR>two.0.CO;2.
- ^ Joe Buchdahl. "Temper, Climate & Environment Information Programme". Ace.mmu.ac.united kingdom of great britain and northern ireland. Archived from the original on 2010-07-01. Retrieved 2012-04-18 .
- ^ Journal of the Atmospheric Sciences (1993). "stratopause". Retrieved 2013-x-18 .
- ^ Barry, R.G.; Chorley, R.J. (1971). Temper, Weather and Climate . London: Menthuen & Co Ltd. p. 65. ISBN9780416079401.
- ^ Tyson, P.D.; Preston-Whyte, R.A. (2013). The Weather condition and Climate of Southern Africa (2nd ed.). Oxford: Oxford University Printing. p. 4.
- ^ "Troposphere". Concise Encyclopedia of Science & Technology. McGraw-Hill. 1984.
It contains almost 4-fifths of the mass of the whole atmosphere.
- ^ "ISS022-Eastward-062672 caption". NASA. Archived from the original on sixteen February 2010. Retrieved 21 September 2012.
- ^ "homosphere – AMS Glossary". Amsglossary.allenpress.com. Archived from the original on 14 September 2010. Retrieved 2010-ten-16 .
- ^ "Earth's Atmosphere". Archived from the original on 2009-06-14.
- ^ "NASA – World Fact Sheet". Nssdc.gsfc.nasa.gov. Archived from the original on 30 October 2010. Retrieved 2010-10-sixteen .
- ^ "Global Surface Temperature Anomalies". Archived from the original on 2009-03-03.
- ^ "Earth'due south Radiation Balance and Oceanic Oestrus Fluxes". Archived from the original on 2005-03-03.
- ^ "Coupled Model Intercomparison Projection Control Run" (PDF). Archived from the original (PDF) on 2008-05-28.
- ^ Geometric altitude vs. temperature, pressure, density, and the speed of sound derived from the 1962 U.Due south. Standard Atmosphere.
- ^ Trenberth, Kevin E.; Smith, Lesley (1970-01-01). "The Mass of the Atmosphere: A Constraint on Global Analyses". Periodical of Climate. 18 (six): 864. Bibcode:2005JCli...18..864T. CiteSeerXx.1.1.727.6573. doi:ten.1175/JCLI-3299.1. S2CID 16754900.
- ^ Lutgens, Frederick K. and Edward J. Tarbuck (1995) The Atmosphere, Prentice Hall, 6th ed., pp. xiv–17, ISBN 0-13-350612-half dozen
- ^ "Atmospheric Temperature Trends, 1979–2005 : Epitome of the Solar day". Earthobservatory.nasa.gov. 2000-01-01. Retrieved 2014-06-10 .
- ^ St. Fleur, Nicholas (19 May 2017). "Spotting Mysterious Twinkles on Earth From a 1000000 Miles Away". The New York Times . Retrieved 20 May 2017.
- ^ Marshak, Alexander; Várnai, Tamás; Kostinski, Alexander (15 May 2017). "Terrestrial glint seen from deep space: oriented ice crystals detected from the Lagrangian point". Geophysical Research Letters. 44 (10): 5197. Bibcode:2017GeoRL..44.5197M. doi:ten.1002/2017GL073248.
- ^ Edlén, Bengt (1966). "The refractive index of air". Metrologia. 2 (2): 71–80. Bibcode:1966Metro...2...71E. doi:10.1088/0026-1394/two/2/002.
- ^ a b Zahnle, K.; Schaefer, L.; Fegley, B. (2010). "Earth'southward Earliest Atmospheres". Cold Bound Harbor Perspectives in Biological science. 2 (10): a004895. doi:ten.1101/cshperspect.a004895. PMC2944365. PMID 20573713.
- ^ B. Windley: The Evolving Continents. Wiley Press, New York 1984
- ^ J. Schopf: Earth's Earliest Biosphere: Its Origin and Evolution. Princeton University Press, Princeton, N.J., 1983
- ^ Timothy W. Lyons, Christopher T. Reinhard & Noah J. Planavsky (2014). "Atmospheric oxygenation iii billion years agone". Nature. 506 (7488): 307–15. Bibcode:2014Natur.506..307L. doi:ten.1038/nature13068. PMID 24553238. S2CID 4443958.
- ^ Martin, Daniel; McKenna, Helen; Livina, Valerie (2016). "The human physiological impact of global deoxygenation". The Periodical of Physiological Sciences. 67 (one): 97–106. doi:x.1007/s12576-016-0501-0. ISSN 1880-6546. PMC5138252. PMID 27848144.
- ^ Graph: Atmospheric Oxygen and CO2 vs Fourth dimension
- ^ Christopher R. Scotese, Dorsum to Earth History : Summary Chart for the Precambrian, Paleomar Project
- ^ Schmidt-Rohr, K. (2020). "Oxygen Is the High-Free energy Molecule Powering Complex Multicellular Life: Central Corrections to Traditional Bioenergetics". ACS Omega 5: 2221-2233. http://dx.doi.org/10.1021/acsomega.9b03352
- ^ Peter Ward:[ane] Out of Thin Air: Dinosaurs, Birds, and Globe'due south Aboriginal Atmosphere
- ^ Starting from [2] Pollution – Definition from the Merriam-Webster Online Dictionary
- ^ IPCC (2021). "Summary for Policymakers" (PDF). IPCC AR6 WG1. pp. four–5. Archived from the original (PDF) on 2021-08-11. Retrieved 2021-xi-20 .
- ^ Northon, Karen (2015-10-nineteen). "Daily Views of Globe Available on New NASA Website". NASA . Retrieved 2015-ten-21 .
External links
| | Wikiquote has quotations related to Air . |
- Interactive global map of current atmospheric and ocean surface conditions.
Source: https://en.wikipedia.org/wiki/Atmosphere_of_Earth
Posted by: frazerearpot.blogspot.com



0 Response to "What 3 Gases Makeup Our Atsophere"
Post a Comment